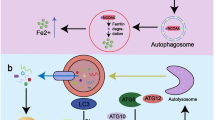
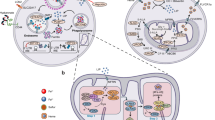
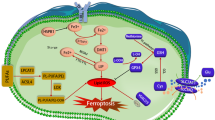
Abstract
Iron metabolism is increasingly recognized as a key player in the development and progression of various cancers. Iron is required for vital cellular processes such as energy production; however, it can also interact with reactive oxygen species to cause cellular toxicity. Consequently, a host of proteins coordinate iron homeostasis, and ferritin stands out as a promising therapeutic target due to its pivotal role in buffering cellular iron levels. This review explores the relevance of ferritin in brain cancers, shedding light on how it influences the biology of both tumor cells and cancer stem cells (CSCs), a population of tumor cells that is notable in their resistance to conventional treatment strategies. Ferritin plays a critical role in protecting against oxidative stress and boosting resistance to ferroptosis, a form of cell death often evaded by CSCs. Development of cutting-edge strategies designed to target ferritin, including ferritinophagy-inducing compounds and novel redox-based therapies that can capitalize on the iron dependency of CSCs is discussed in context. We propose that the iron addiction of brain cancer cells provides a specific susceptibility, whereby removing their iron buffering mechanism via targeting of ferritin can result in favorable treatment outcomes, including the induction of iron-dependent cell death. Future studies on the modulation of ferritin offer a ground-breaking therapeutic strategy to undermine CSC-driven tumor growth, overcome resistance to conventional therapies, and ultimately improve treatment outcomes for patients battling brain cancers.
Similar content being viewed by others
References
Chen Y, Fan Z, Yang Y, Gu C. Iron metabolism and its contribution to cancer (Review). Int J Oncol. 2019. http://www.spandidos-publications.com/10.3892/ijo.2019.4720
Rodriguez R, Schreiber SL, Conrad M. Persister cancer cells: Iron addiction and vulnerability to ferroptosis. Mol Cell. 2022;82:728–40.
Schonberg DL, Miller TE, Wu Q, Flavahan WA, Das NK, Hale JS, et al. Preferential iron trafficking characterizes glioblastoma stem-like cells. Cancer Cell. 2015;28:441–55.
Dixon SJ, Lemberg KM, Lamprecht MR, Skouta R, Zaitsev EM, Gleason CE, et al. Ferroptosis: an iron-dependent form of nonapoptotic cell death. Cell. 2012;149:1060–72.
Fenton HJH. Oxidation of tartaric acid in presence of iron. University of Windsor. 1894. p. 11.
Zhu ZY, Liu YD, Gong Y, Jin W, Topchiy E, Turdi S, et al. Mitochondrial aldehyde dehydrogenase (ALDH2) rescues cardiac contractile dysfunction in an APP/PS1 murine model of Alzheimer’s disease via inhibition of ACSL4-dependent ferroptosis. Acta Pharmacol Sin. 2022;43:39–49.
Yang WS, Stockwell BR. Ferroptosis: death by lipid peroxidation. Trends Cell Biol. 2016;26:165–76.
Von Krusenstiern AN, Robson RN, Qian N, Qiu B, Hu F, Reznik E, et al. Identification of essential sites of lipid peroxidation in ferroptosis. Nat Chem Biol. 2023;19:719–30.
Wang P, Yao Q, Zhu D, Yang X, Chen Q, Lu Q, et al. Resveratrol protects against deoxynivalenol-induced ferroptosis in HepG2 cells. Toxicology. 2023;494:153589.
Geng N, Shi B-J, Li S-L, Zhong Z-Y, Li Y-C, Xua W-L, et al. Knockdown of ferroportin accelerates erastin-induced ferroptosis in neuroblastoma cells. Eur Rev Med Pharmacol Sci. 2018;22:3826–36.
Dolma S, Lessnick SL, Hahn WC, Stockwell BR. Identification of genotype-selective antitumor agents using synthetic lethal chemical screening in engineered human tumor cells. Cancer Cell. 2003;3:285–96.
Yang WS, Stockwell BR. Synthetic lethal screening identifies compounds activating iron-dependent, nonapoptotic cell death in oncogenic-RAS-harboring cancer cells. Chem Biol. 2008;15:234–45.
Mo Q, Song C, Hua Y, Wang W, Liu A. CYP2E1 mediated deoxynivalenol-induced hepatocyte toxicity by regulating ferroptosis. Toxicology. 2024;508:153923.
Zhou X, Wang H, Yan B, Nie X, Chen Q, Yang X, et al. Ferroptosis in cardiovascular diseases and ferroptosis-related intervention approaches. Cardiovasc Drugs Ther. 2024. https://link.springer.com/10.1007/s10557-024-07642-5.
Ibrahim O, O’Sullivan J. Iron chelators in cancer therapy. BioMetals. 2020;33:201–15.
Wallace DF. The regulation of iron absorption and homeostasis. Clin Biochem Rev. 2016;37:51–62.
Gao T, O’Brian MR. Iron-dependent cytochrome c 1 expression is mediated by the status of heme in Bradyrhizobium japonicum. J Bacteriol. 2005;187:5084–9.
Mühlenhoff U, Richhardt N, Gerber J, Lill R. Characterization of iron–sulfur protein assembly in isolated mitochondria. J Biol Chem. 2002;277:29810–6.
Read AD, Bentley RE, Archer SL, Dunham-Snary KJ. Mitochondrial iron–sulfur clusters: structure, function, and an emerging role in vascular biology. Redox Biol. 2021;47:102164.
Rangaraju V, Calloway N, Ryan TA. Activity-driven kocal ATP synthesis is required for synaptic function. Cell. 2014;156:825–35.
Pathak D, Shields LY, Mendelsohn BA, Haddad D, Lin W, Gerencser AA, et al. The role of mitochondrially derived ATP in synaptic vesicle recycling. J Biol Chem. 2015;290:22325–36.
Van Bergen JMG, Li X, Hua J, Schreiner SJ, Steininger SC, Quevenco FC, et al. Colocalization of cerebral iron with amyloid beta in mild cognitive impairment. Sci Rep. 2016;6:35514.
Yamamoto A, Shin R, Hasegawa K, Naiki H, Sato H, Yoshimasu F, et al. Iron (III) induces aggregation of hyperphosphorylated τ and its reduction to iron (II) reverses the aggregation: implications in the formation of neurofibrillary tangles of Alzheimer’s disease. J Neurochem. 2002;82:1137–47.
Kostka M, Högen T, Danzer KM, Levin J, Habeck M, Wirth A, et al. Single particle characterization of iron-induced pore-forming α-synuclein oligomers. J Biol Chem. 2008;283:10992–1003.
Levin J, Högen T, Hillmer AS, Bader B, Schmidt F, Kamp F, et al. Generation of ferric iron links oxidative stress to α-synuclein oligomer formation. J Park Dis. 2011;1:205–16.
Kumar N, Rizek P, Jog M. Neuroferritinopathy: pathophysiology, presentation, differential diagnoses and management. Tremor Hyperkinet Mov. 2016;6:355.
Li Y, Guan Q, Chen Y, Han H, Liu W, Nie Z. Transferrin receptor and ferritin-H are developmentally regulated in oligodendrocyte lineage cells. Neural Regen Res. 2013;8:6.
Chang S, Wang P, Han Y, Ma Q, Liu Z, Zhong S, et al. Ferrodifferentiation regulates neurodevelopment via ROS generation. Sci China Life Sci. 2023;66:1841–57.
Pérez MJ, Carden TR, Dos Santos Claro PA, Silberstein S, Páez PM, Cheli VT, et al. Transferrin enhances neuronal differentiation. ASN Neuro. 2023;15:17590914231170703.
Ma Z, Wang W, Yang X, Rui M, Wang S. Glial ferritin maintains neural stem cells via transporting iron required for self-renewal in Drosophila. Elife. 2024. https://elifesciences.org/reviewed-preprints/93604v2.
Ding Y, Yao S, Guo S, Meng W, Li J, Wang F, et al. Ferroportin 1 depletion in neural stem cells promotes hippocampal neurogenesis and cognitive function in mice. Pharmacol Res. 2025;216:107778.
McCann S, Perapoch Amadó M, Moore SE. The role of iron in brain development: a systematic review. Nutrients. 2020;12:2001.
Beard J. Iron deficiency alters brain development and functioning. J Nutr. 2003;133:1468S–1472S.
Tang C, Yang J, Zhu C, Ding Y, Yang S, Xu B, et al. Iron metabolism disorder and multiple sclerosis: a comprehensive analysis. Front Immunol. 2024;15:1376838.
Lawson DM, Treffry A, Artymiuk PJ, Harrison PM, Yewdall SJ, Luzzago A, et al. Identification of the ferroxidase centre in ferritin. FEBS Lett. 1989;254:207–10.
Arosio P, Ingrassia R, Cavadini P. Ferritins: a family of molecules for iron storage, antioxidation and more. Biochim Biophys Acta BBA—Gen Subj. 2009;1790:589–99.
Toyokuni S. Role of iron in carcinogenesis: cancer as a ferrotoxic disease. Cancer Sci. 2009;100:9–16.
Nilsson R, Liu NA. Nuclear DNA damages generated by reactive oxygen molecules (ROS) under oxidative stress and their relevance to human cancers, including ionizing radiation-induced neoplasia part II: Relation between ROS-induced DNA damages and human cancer. Radiat Med Prot. 2020;1:196–204.
Zhao H, Wu L, Yan G, Chen Y, Zhou M, Wu Y, et al. Inflammation and tumor progression: signaling pathways and targeted intervention. Signal Transduct Target Ther. 2021;6:263.
Zeidan RS, Yoon HS, Yang JJ, Sobh A, Braithwaite D, Mankowski R, et al. Iron and cancer: overview of the evidence from population-based studies. Front Oncol. 2024;14. https://www.frontiersin.org/articles/10.3389/fonc.2024.1393195/full.
Stevens RG, Jones DY, Micozzi MS, Taylor PR. Body iron stores and the risk of cancer. N Engl J Med. 1988;319:1047–52.
Selby JV, Friedman GD. Epidemiologic evidence of an association between body iron stores and risk of cancer. Int J Cancer. 1988;41:677–82.
Stevens RG, Beasley P, Blumberg BS. Iron-binding proteins and risk of cancer in Taiwan. J Natl Cancer Inst. 1986;76:605–10.
Magnusson G, Flodh H, Malmfors T. Oncological Study in Rats of Ferastral®, an Iron–poly-(sorbitol-gluconic acid) complex, after Intramuscular administration. Scand J Haematol. 1977;19:87–98.
Golberg L, Martin LE, Smith JP. Iron overloading phenomena in animals. Toxicol Appl Pharmacol. 1960;2:683–707.
Shi X, Zhang A, Lu J, Wang X, Yi C, Yang F. An overview of heavy chain ferritin in cancer. Front Biosci-Landmark. 2023;28:182.
Weinstein RE, Bond BH, Silberberg BK, Vaughn CB, Subbaiah P, Pieper DR. Tissue ferritin concentration and prognosis in carcinoma of the breast. Breast Cancer Res Treat. 1989;14:349–53.
Ji M, Li XD, Shi HB, Ning ZH, Zhao WQ, Wang Q, et al. Clinical significance of serum ferritin in elderly patients with primary lung carcinoma. Tumor Biol. 2014;35:10195–9.
Wang J, Liu QX, Teng DL, Ding YB, Lu GT, Gong WJ, et al. Elevated serum ferritin levels are associated with severity and prognosis of severe acute pancreatitis: a preliminary cohort study. BMC Gastroenterol. 2022;22:408.
Feng Z, Chen JW, Feng JH, Shen F, Cai WS, Cao J, et al. The association between serum ferritin and colorectal cancer. Int J Clin Exp Med. 2015;8:22293–99.
Mortazavi H, Sabour S, Baharvand M, Manifar S, Akkafan R. Serum levels of ferritin, copper, and zinc in patients with oral cancer. Biomed J. 2014;37:331.
Kalousová M, Krechler T, Jáchymová M, Kuběna AA, Žák A, Zima T. Ferritin as an independent mortality predictor in patients with pancreas cancer. Results of a pilot study. Tumor Biol. 2012;33:1695–700.
Moroz V, Machin D, Hero B, Ladenstein R, Berthold F, Kao P, et al. The prognostic strength of serum LDH and serum ferritin in children with neuroblastoma: a report from the International Neuroblastoma Risk Group (INRG) project. Pediatr Blood Cancer. 2020;67:e28359.
Wang X, An P, Zeng J, Liu X, Wang B, Fang X, et al. Serum ferritin in combination with prostate-specific antigen improves predictive accuracy for prostate cancer. Oncotarget. 2017;8:17862–72.
Lorenzi M, Lorenzi B, Vernillo R. Serum ferritin in colorectal cancer patients and its prognostic evaluation. Int J Biol Markers. 2006;21:235–41.
Wu SJ, Zhang ZZ, Cheng NS, Xiong XZ, Yang L. Preoperative serum ferritin is an independent prognostic factor for liver cancer after hepatectomy. Surg Oncol. 2019;29:159–67.
Wang SL, Cao S, Wu R, Chi F, Tang MY, Jin XY, et al. Serum ferritin predicted prognosis in patients with locally advanced pancreatic cancer. Future Oncol. 2015;11:2905–10.
Rychtarcikova Z, Lettlova S, Tomkova V, Korenkova V, Langerova L, Simonova E, et al. Tumor-initiating cells of breast and prostate origin show alterations in the expression of genes related to iron metabolism. Oncotarget. 2017;8:6376–98.
Kindrat I, Tryndyak V, De Conti A, Shpyleva S, Mudalige TK, Kobets T, et al. MicroRNA-152-mediated dysregulation of hepatic transferrin receptor 1 in liver carcinogenesis. Oncotarget. 2016;7:1276–87.
Basuli D, Tesfay L, Deng Z, Paul B, Yamamoto Y, Ning G, et al. Iron addiction: a novel therapeutic target in ovarian cancer. Oncogene. 2017;36:4089–99.
Wang B, Zhang J, Song F, Tian M, Shi B, Jiang H, et al. EGFR regulates iron homeostasis to promote cancer growth through redistribution of transferrin receptor 1. Cancer Lett. 2016;381:331–40.
Xue X, Ramakrishnan SK, Weisz K, Triner D, Xie L, Attili D, et al. Iron uptake via DMT1 integrates cell cycle with JAK-STAT3 signaling to promote colorectal tumorigenesis. Cell Metab. 2016;24:447–61.
Gu Z, Wang H, Xia J, Yang Y, Jin Z, Xu H, et al. Decreased ferroportin promotes myeloma cell growth and osteoclast differentiation. Cancer Res. 2015;75:2211–21.
Feng G, Arima Y, Midorikawa K, Kobayashi H, Oikawa S, Zhao W, et al. Knockdown of TFRC suppressed the progression of nasopharyngeal carcinoma by downregulating the PI3K/Akt/mTOR pathway. Cancer Cell Int. 2023;23:185.
Kim H, Villareal LB, Liu Z, Haneef M, Falcon DM, Martin DR, et al. Transferrin receptor-mediated iron uptake promotes colon tumorigenesis. Adv Sci. 2023;10:2207693.
Hann HWL, Stahlhut MW, Menduke H. Iron enhances tumor growth. Observation on spontaneous mammary tumors in mice. Cancer. 1991;68:2407–10. Dec 1.
Hann HW, Stahlhut MW, Blumberg BS. Iron nutrition and tumor growth: decreased tumor growth in iron-deficient mice. Cancer Res. 1988;48:4168–70.
Lui GYL, Obeidy P, Ford SJ, Tselepis C, Sharp DM, Jansson PJ, et al. The iron chelator, deferasirox, as a novel strategy for cancer treatment: oral activity against human lung tumor xenografts and molecular mechanism of action. Mol Pharmacol. 2013;83:179–90.
White S, Taetle R, Seligman PA, Rutherford M, Trowbridge IS. Combinations of anti-transferrin receptor monoclonal antibodies inhibit human tumor cell growth in vitro and in vivo: evidence for synergistic antiproliferative effects. Cancer Res. 1990;50:6295–301.
Belvin BR, Lewis JP. Ferroportin depletes iron needed for cell cycle progression in head and neck squamous cell carcinoma. Front Oncol. 2023;12:1025434.
Tian T, Li X, Zhang J. mTOR signaling in cancer and mTOR inhibitors in solid tumor targeting therapy. Int J Mol Sci. 2019;20:755.
Hanahan D. Hallmarks of cancer: new dimensions. Cancer Discov. 2022;12:31–46.
Tee AR, Fingar DC, Manning BD, Kwiatkowski DJ, Cantley LC, Blenis J. Tuberous sclerosis complex-1 and -2 gene products function together to inhibit mammalian target of rapamycin (mTOR)-mediated downstream signaling. Proc Natl Acad Sci USA. 2002;99:13571–6.
Saxton RA, Sabatini DM. mTOR signaling in growth, metabolism, and disease. Cell. 2017;168:960–76.
Shang C, Zhou H, Liu W, Shen T, Luo Y, Huang S. Iron chelation inhibits mTORC1 signaling involving activation of AMPK and REDD1/Bnip3 pathways. Oncogene. 2020;39:5201–13.
Kim DH, Kim JH, Kim EH, Na HK, Cha YN, Chung JH, et al. 15-Deoxy-Δ 12,14 -prostaglandin J 2 upregulates the expression of heme oxygenase-1 and subsequently matrix metalloproteinase-1 in human breast cancer cells: possible roles of iron and ROS. Carcinogenesis. 2009;30:645–54.
Cheng M, Liu P, Xu LX. Iron promotes breast cancer cell migration via IL-6/JAK2/STAT3 signaling pathways in a paracrine or autocrine IL-6-rich inflammatory environment. J Inorg Biochem. 2020;210:111159.
Mehta KJ, Sharp PA. Iron elevates mesenchymal and metastatic biomarkers in HepG2 cells. Sci Rep. 2020;10:21926.
Bhutia YD, Ogura J, Grippo PJ, Torres C, Sato T, Wachtel M, et al. Chronic exposure to excess iron promotes EMT and cancer via p53 loss in pancreatic cancer. Asian J Pharm Sci. 2020;15:237–51.
Chen Z, Zhang D, Yue F, Zheng M, Kovacevic Z, Richardson DR. The Iron Chelators Dp44mT and DFO inhibit TGF-β-induced epithelial–mesenchymal transition via up-regulation of N-Myc downstream-regulated gene 1 (NDRG1). J Biol Chem. 2012;287:17016–28.
Le NTV, Richardson DR. Iron chelators with high antiproliferative activity up-regulate the expression of a growth inhibitory and metastasis suppressor gene: a link between iron metabolism and proliferation. Blood. 2004;104:2967–75.
Oft M, Heider KH, Beug H. TGFβ signaling is necessary for carcinoma cell invasiveness and metastasis. Curr Biol. 1998;8:1243–52.
Recalcati S, Locati M, Marini A, Santambrogio P, Zaninotto F, De Pizzol M, et al. Differential regulation of iron homeostasis during human macrophage polarized activation. Eur J Immunol. 2010;40:824–35.
Li H, Yang C, Wei Y, Li X, Jiang W, Xu Y, et al. Ferritin light chain promotes the reprogramming of glioma immune microenvironment and facilitates glioma progression. Theranostics. 2023;13:3794–813.
Ermakov MS, Nushtaeva AA, Richter VA, Koval OA. Cancer-associated fibroblasts and their role in tumor progression. Vavilov J Genet Breed. 2022;26:14–21.
Zhang K, Liu K, Hu B, Du G, Chen X, Xiao L, et al. Iron-loaded cancer-associated fibroblasts induce immunosuppression in prostate cancer. Nat Commun. 2024;15:9050.
Parks SK, Cormerais Y, Pouysségur J. Hypoxia and cellular metabolism in tumour pathophysiology. J Physiol. 2017;595:2439–50.
Torti SV, Torti FM. Iron and cancer: more ore to be mined. Nat Rev Cancer. 2013;13:342–55.
Kaelin WG, Ratcliffe PJ. Oxygen sensing by metazoans: the central role of the HIF hydroxylase pathway. Mol Cell. 2008;30:393–402.
Eckard J, Dai J, Wu J, Jian J, Yang Q, Chen H, et al. Effects of cellular iron deficiency on the formation of vascular endothelial growth factor and angiogenesis. Cancer Cell Int. 2010;10:28.
Plaks V, Kong N, Werb Z. The cancer stem cell niche: how essential is the niche in regulating stemness of tumor cells? Cell Stem Cell. 2015;16:225–38.
Zhang Q, Han Z, Zhu Y, Chen J, Li W. Role of hypoxia inducible factor-1 in cancer stem cells (review). Mol Med Rep. 2020;23:1–1.
Batlle E, Clevers H. Cancer stem cells revisited. Nat Med. 2017;23:1124–34.
Yu R, Hang Y, Tsai HI, Wang D, Zhu H. Iron metabolism: backfire of cancer cell stemness and therapeutic modalities. Cancer Cell Int. 2024;24:157.
Song Q, Peng S, Sun Z, Heng X, Zhu X. Temozolomide drives ferroptosis via a DMT1-dependent pathway in glioblastoma cells. Yonsei Med J. 2021;62:843.
Ninomiya T, Ohara T, Noma K, Katsura Y, Katsube R, Kashima H, et al. Iron depletion is a novel therapeutic strategy to target cancer stem cells. Oncotarget. 2017;8:98405–16.
Stuelten CH, Mertins SD, Busch JI, Gowens M, Scudiero DA, Burkett MW, et al. Complex display of putative tumor stem cell markers in the NCI60 tumor cell line panel. Stem Cells. 2010;28:649–60.
Müller S, Sindikubwabo F, Cañeque T, Lafon A, Versini A, Lombard B, et al. CD44 regulates epigenetic plasticity by mediating iron endocytosis. Nat Chem. 2020;12:929–38.
Bourseau-Guilmain E, Griveau A, Benoit JP, Garcion E. The importance of the stem cell marker prominin-1/CD133 in the uptake of transferrin and in iron metabolism in human colon cancer Caco-2 cells. PLoS ONE. 2011;6:e25515.
Raggi C, Gammella E, Correnti M, Buratti P, Forti E, Andersen JB, et al. Dysregulation of iron metabolism in cholangiocarcinoma stem-like cells. Sci Rep. 2017;7:17667.
Mai TT, Hamaï A, Hienzsch A, Cañeque T, Müller S, Wicinski J, et al. Salinomycin kills cancer stem cells by sequestering iron in lysosomes. Nat Chem. 2017;9:1025–33.
Conti L, Lanzardo S, Ruiu R, Cadenazzi M, Cavallo F, Aime S, et al. L-Ferritin targets breast cancer stem cells and delivers therapeutic and imaging agents. Oncotarget. 2016;7:66713–27.
Xiao C, Fu X, Wang Y, Liu H, Jiang Y, Zhao Z, et al. Transferrin receptor regulates malignancies and the stemness of hepatocellular carcinoma-derived cancer stem-like cells by affecting iron accumulation. PLoS ONE. 2020;15:e0243812.
Baksh SC, Finley LWS. Metabolic coordination of cell fate by α-ketoglutarate-dependent dioxygenases. Trends Cell Biol. 2021;31:24–36.
Han S, Liu Y, Cai SJ, Qian M, Ding J, Larion M, et al. IDH mutation in glioma: molecular mechanisms and potential therapeutic targets. Br J Cancer. 2020;122:1580–9.
Ambrosini G, Cordani M, Zarrabi A, Alcon-Rodriguez S, Sainz RM, Velasco G, et al. Transcending frontiers in prostate cancer: the role of oncometabolites on epigenetic regulation, CSCs, and tumor microenvironment to identify new therapeutic strategies. Cell Commun Signal. 2024;22:36.
Xu W, Yang H, Liu Y, Yang Y, Wang P, Kim SH, et al. Oncometabolite 2-hydroxyglutarate is a competitive inhibitor of α-ketoglutarate-dependent dioxygenases. Cancer Cell. 2011;19:17–30.
Xiao M, Yang H, Xu W, Ma S, Lin H, Zhu H, et al. Inhibition of α-KG-dependent histone and DNA demethylases by fumarate and succinate that are accumulated in mutations of FH and SDH tumor suppressors. Genes Dev. 2012;26:1326–38.
Young JI, Züchner S, Wang G. Regulation of the epigenome by vitamin C. Annu Rev Nutr. 2015;35:545–64.
Withey K, Brougham MFH, Paciarotti I, McKenzie JM, Wilson DC, Revuelta Iniesta R. Associations of ferritin and folate status with clinical outcomes in childhood cancer patients: a Prospective Cohort study. Pediatr Blood Cancer. 2025;72:e31645.
Jaksch-Bogensperger H, Spiegl-Kreinecker S, Arosio P, Eckl P, Golaszewski S, Ebner Y, et al. Ferritin in glioblastoma. Br J Cancer. 2020;122:1441–4.
Schwartzbaum JA, Cornwell DG. Oxidant stress and glioblastoma multiforme risk: serum antioxidants, γ-glutamyl transpeptidase, and ferritin. Nutr Cancer. 2000;38:40–9.
Fisher J, Devraj K, Ingram J, Slagle-Webb B, Madhankumar AB, Liu X, et al. Ferritin: a novel mechanism for delivery of iron to the brain and other organs. Am J Physiol-Cell Physiol. 2007;293:C641–9.
Wu T, Li Y, Liu B, Zhang S, Wu L, Zhu X, et al. Expression of ferritin light chain (FTL) is elevated in glioblastoma, and FTL silencing inhibits glioblastoma cell proliferation via the GADD45/JNK pathway. PLoS ONE. 2016;11:e0149361.
Yang FC, Wang C, Zhu J, Gai QJ, Mao M, He J, et al. Inhibitory effects of temozolomide on glioma cells is sensitized by RSL3-induced ferroptosis but negatively correlated with expression of ferritin heavy chain 1 and ferritin light chain. Lab Investig. 2022;102:741–52.
Rosager AM, Sørensen MD, Dahlrot RH, Hansen S, Schonberg DL, Rich JN, et al. Transferrin receptor-1 and ferritin heavy and light chains in astrocytic brain tumors: expression and prognostic value. PLoS ONE. 2017;12:e0182954.
Chekhun VF, Lukianova NY, Chekhun SV, Bezdieniezhnykh NO, Zadvorniy TV, Borikun TV, et al. Association of CD44+CD24−/low with markers of aggressiveness and plasticity of cell lines and tumors of patients with breast cancer. Exp Oncol. 2017;39:203–11.
Kanojia D, Zhou W, Zhang J, Jie C, Lo P, Wang Q, et al. Proteomic profiling of cancer stem cells derived from primary tumors of HER2/Neu transgenic mice. Proteomics. 2012;12:3407–15.
Gong AH, Wei P, Zhang S, Yao J, Yuan Y, Zhou AD, et al. FoxM1 drives a feed-forward STAT3-activation signaling loop that promotes the self-renewal and tumorigenicity of glioblastoma stem-like cells. Cancer Res. 2015;75:2337–48.
Ravi V, Madhankumar AB, Abraham T, Slagle-Webb B, Connor JR. Liposomal delivery of ferritin heavy chain 1 (FTH1) siRNA in patient xenograft derived glioblastoma initiating cells suggests different sensitivities to radiation and distinct survival mechanisms. PLoS ONE. 2019;14:e0221952.
Lobello N, Biamonte F, Pisanu ME, Faniello MC, Jakopin Ž, Chiarella E, et al. Ferritin heavy chain is a negative regulator of ovarian cancer stem cell expansion and epithelial to mesenchymal transition. Oncotarget. 2016;7:62019–33.
Aversa I, Zolea F, Ieranò C, Bulotta S, Trotta AM, Faniello MC, et al. Epithelial-to-mesenchymal transition in FHC-silenced cells: the role of CXCR4/CXCL12 axis. J Exp Clin Cancer Res. 2017;36:104.
Lei ZN, Teng QX, Koya J, Liu Y, Chen Z, Zeng L, et al. The correlation between cancer stem cells and epithelial-mesenchymal transition: molecular mechanisms and significance in cancer theragnosis. Front Immunol. 2024;15:1417201.
Zhang KH, Tian HY, Gao X, Lei WW, Hu Y, Wang DM, et al. Ferritin heavy chain-mediated iron homeostasis and subsequent increased reactive oxygen species production are essential for epithelial–mesenchymal transition. Cancer Res. 2009;69:5340–8.
Sioutas A, Vainikka L, Kentson M, Dam Larsen S, Wennerström U, Jacobson P, et al. Oxidant-induced autophagy and ferritin degradation contribute to epithelial– mesenchymal transition through lysosomal iron. J Inflamm Res. 2017;10:29–39.
Shesh B, Slagle-Webb B, Shenoy G, Khristov V, Zacharia BE, Connor JR. Uptake of H-ferritin by glioblastoma stem cells and its impact on their invasion capacity. J Cancer Res Clin Oncol. 2023;149:9691–703.
Gerlee P, Nelander S. The impact of phenotypic switching on glioblastoma growth and invasion. PLoS Comput Biol. 2012;8:e1002556.
Hatzikirou H, Basanta D, Simon M, Schaller K, Deutsch A. Go or Grow’: the key to the emergence of invasion in tumour progression? Math Med Biol. 2012;29:49–65.
Lei G, Zhuang L, Gan B. The roles of ferroptosis in cancer: tumor suppression, tumor microenvironment, and therapeutic interventions. Cancer Cell. 2024;42:513–34.
Wang J, Wu N, Peng M, Oyang L, Jiang X, Peng Q, et al. Ferritinophagy: research advance and clinical significance in cancers. Cell Death Discov. 2023;9:463.
Fang Y, Chen X, Tan Q, Zhou H, Xu J, Gu Q. Inhibiting ferroptosis through disrupting the NCOA4–FTH1 interaction: a new mechanism of action. ACS Cent Sci. 2021;7:980–9.
Zhang Y, Kong Y, Ma Y, Ni S, Wikerholmen T, Xi K, et al. Loss of COPZ1 induces NCOA4 mediated autophagy and ferroptosis in glioblastoma cell lines. Oncogene. 2021;40:1425–39.
Li K, Chen B, Xu A, Shen J, Li K, Hao K, et al. TRIM7 modulates NCOA4-mediated ferritinophagy and ferroptosis in glioblastoma cells. Redox Biol. 2022;56:102451.
Hayashima K, Kimura I, Katoh H. Role of ferritinophagy in cystine deprivation-induced cell death in glioblastoma cells. Biochem Biophys Res Commun. 2021;539:56–63.
Daher B, Vučetić M, Pouysségur J. Cysteine depletion, a key action to challenge cancer cells to ferroptotic cell death. Front Oncol. 2020;10:723.
Daher B, Parks SK, Durivault J, Cormerais Y, Baidarjad H, Tambutte E, et al. Genetic ablation of the cystine transporter xCT in PDAC cells inhibits mTORC1, growth, survival, and tumor formation via nutrient and oxidative stresses. Cancer Res. 2019;79:3877–90.
Wu W, Xu C, Ling X, Fan C, Buckley BP, Chernov MV, et al. Targeting RING domains of Mdm2–MdmX E3 complex activates apoptotic arm of the p53 pathway in leukemia/lymphoma cells. Cell Death Dis. 2015;6:e2035–e2035.
Li J, Lama R, Galster SL, Inigo JR, Wu J, Chandra D, et al. Small-Molecule MMRi62 induces ferroptosis and inhibits metastasis in pancreatic cancer via degradation of ferritin heavy chain and mutant p53. Mol Cancer Ther. 2022;21:535–45.
Lama R, Galster SL, Xu C, Davison LW, Chemler SR, Wang X. Dual Targeting of MDM4 and FTH1 by MMRi71 for induced protein degradation and p53-independent apoptosis in leukemia cells. Molecules. 2022;27:7665 Nov 8.
Mussa A, Mohd Idris RA, Ahmed N, Ahmad S, Murtadha AH, Tengku Din TADAA, et al. High-dose vitamin C for cancer therapy. Pharmaceuticals. 2022;15:711.
Baillie N, Carr AC, Peng S. The use of intravenous vitamin C as a supportive therapy for a patient with glioblastoma multiforme. Antioxidants. 2018;7:115.
Wang X, Xu S, Zhang L, Cheng X, Yu H, Bao J, et al. Vitamin C induces ferroptosis in anaplastic thyroid cancer cells by ferritinophagy activation. Biochem Biophys Res Commun. 2021;551:46–53.
Chevignard M, Câmara-Costa H, Doz F, Dellatolas G. Core deficits and quality of survival after childhood medulloblastoma: a review. Neuro-Oncol Pract. 2017;4:82–97.
Baidoo KE, Yong K, Brechbiel MW. Molecular pathways: targeted α-particle radiation therapy. Clin Cancer Res. 2013;19:530–7.
Gotorbe C, Segui F, Echavidre W, Durivault J, Blanchard T, Vial V, et al. Exploiting integrin-αVβ3 to enhance radiotherapy efficacy in medulloblastoma via ferroptosis. Curr Oncol. 2024;31:7390–402.
Lei G, Zhang Y, Koppula P, Liu X, Zhang J, Lin SH, et al. The role of ferroptosis in ionizing radiation-induced cell death and tumor suppression. Cell Res. 2020;30:146–62.
Ye LF, Chaudhary KR, Zandkarimi F, Harken AD, Kinslow CJ, Upadhyayula PS, et al. Radiation-induced lipid peroxidation triggers ferroptosis and synergizes with ferroptosis inducers. ACS Chem Biol. 2020;15:469–84.
Olivares-Urbano MA, Griñán-Lisón C, Marchal JA, Núñez MI. CSC radioresistance: a therapeutic challenge to improve radiotherapy effectiveness in cancer. Cells. 2020;9:1651.
Caverzan MD, Ibarra LE. Advancing glioblastoma treatment through iron metabolism: a focus on TfR1 and Ferroptosis innovations. Int J Biol Macromol. 2024;278:134777.
Loftus LV, Rolle LTA, Wang B, Pienta KJ, Amend SR. Dysregulation of labile iron predisposes chemotherapy resistant cancer cells to ferroptosis. Int J Mol Sci. 2025;26:4193.
Liu X, Madhankumar AB, Slagle-Webb B, Sheehan JM, Surguladze N, Connor JR. Heavy chain ferritin siRNA delivered by cationic liposomes increases sensitivity of cancer cells to chemotherapeutic agents. Cancer Res. 2011;71:2240–9.
Kazan HH, Urfali-Mamatoglu C, Gunduz U. Iron metabolism and drug resistance in cancer. BioMetals. 2017;30:629–41.
Mokarram N, Case A, Hossainy NN, Lyon JG, MacDonald TJ, Bellamkonda R. Device-assisted strategies for drug delivery across the blood-brain barrier to treat glioblastoma. Commun Mater. 2025;6:5.
Drechsel DA, Patel M. Role of reactive oxygen species in the neurotoxicity of environmental agents implicated in Parkinson’s disease. Free Radic Biol Med. 2008;44:1873–86.
Levi S, Ripamonti M, Moro AS, Cozzi A. Iron imbalance in neurodegeneration. Mol Psychiatry. 2024;29:1139–52.
Cameron E, Campbell A. The orthomolecular treatment of cancer II. Clinical trial of high-dose ascorbic acid supplements in advanced human cancer. Chem Biol Interact. 1974;9:285–315.
Cameron E, Pauling L. Supplemental ascorbate in the supportive treatment of cancer: prolongation of survival times in terminal human cancer. Proc Natl Acad Sci USA. 1976;73:3685–9.
Chen Y, Li W, Kwon S, Wang Y, Li Z, Hu Q. Small-molecule ferritin degrader as a pyroptosis inducer. J Am Chem Soc. 2023;145:9815–24.
Qu C, Dai E, Lai T, Cao G, Liu J, Kang R, et al. Itaconic acid induces ferroptosis by activating ferritinophagy. Biochem Biophys Res Commun. 2021;583:56–62.
Sui S, Zhang J, Xu S, Wang Q, Wang P, Pang D. Ferritinophagy is required for the induction of ferroptosis by the bromodomain protein BRD4 inhibitor (+)-JQ1 in cancer cells. Cell Death Dis. 2019;10:331.
Huang T, Sun Y, Li Y, Wang T, Fu Y, Li C, et al. Growth inhibition of a novel iron chelator, dpdtc, against hepatoma carcinoma cell lines partly attributed to ferritinophagy-mediated lysosomal ROS generation. Oxid Med Cell Longev. 2018;2018:4928703.
Xiu Z, Zhu Y, Han J, Li Y, Yang X, Yang G, et al. Caryophyllene oxide induces ferritinophagy by regulating the NCOA4/FTH1/LC3 pathway in hepatocellular carcinoma. Front Pharmacol. 2022;13:930958.
Wang Y, Kuang S, Li K, Chen S, Yang M, Deng K, et al. Astragalin promotes HSCs ferroptosis through NCOA4 mediated ferritinophagy to alleviate liver fibrosis in zebrafish and mice. Commun Biol. 2025;8:1081.
Du J, Wang T, Li Y, Zhou Y, Wang X, Yu X, et al. DHA inhibits proliferation and induces ferroptosis of leukemia cells through autophagy dependent degradation of ferritin. Free Radic Biol Med. 2019;131:356–69.
Badu-Boateng C, Naftalin RJ. Ascorbate and ferritin interactions: Consequences for iron release in vitro and in vivo and implications for inflammation. Free Radic Biol Med. 2019;133:75–87.
Zhu L, You Y, Zhu M, Song Y, Zhang J, Hu J, et al. Ferritin-hijacking nanoparticles spatiotemporally directing endogenous ferroptosis for synergistic anticancer therapy. Adv Mater. 2022;34:2207174.
Acknowledgements
This work was fully supported by the Government of Monaco, including a PhD thesis (FS).
Author information
Authors and Affiliations
Contributions
Conception and design: M. Vucetic, V. Picco, and F. Segui. Writing, review, and/or revision of the manuscript: M. Vucetic, V. Picco, F. Segui, and S.K. Parks. Study supervision: M. Vucetic and V. Picco.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Edited by Professor Franscesca Pentimalli
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Segui, F., Parks, S.K., Vucetic, M. et al. A fistful of iron: ferritin as a vulnerability point of the brain cancers. Cell Death Dis (2026). https://doi.org/10.1038/s41419-026-08564-w
Received:
Revised:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41419-026-08564-w