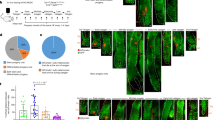
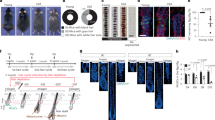
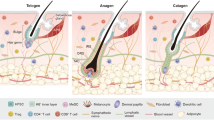
Abstract
Melanocyte stem cells (McSCs) are a crucial melanocyte reservoir within the hair follicle niche. This review provides an overview of the processes for McSC quiescence and activation. Because McSCs closely interact with hair follicle stem cells, we have focused on this interaction. Given the high prevalence of hair graying, the McSC system serves as a model for cellular aging. Here, we highlight current research on the mechanisms of hair graying.
Similar content being viewed by others
References
Morrison, S. J. & Spradling, A. C. Stem cells and niches: mechanisms that promote stem cell maintenance throughout life. Cell 132, 598–611 (2008).
Cotsarelis, G., Sun, T. T. & Lavker, R. M. Label-retaining cells reside in the bulge area of pilosebaceous unit: implications for follicular stem cells, hair cycle, and skin carcinogenesis. Cell 61, 1329–1337 (1990).
Lin, J. Y. & Fisher, D. E. Melanocyte biology and skin pigmentation. Nature 445, 843–850 (2007).
Mort, R. L., Jackson, I. J. & Patton, E. E. The melanocyte lineage in development and disease. Development 142, 620–632 (2015).
Nishimura, E. K. Melanocyte stem cells: a melanocyte reservoir in hair follicles for hair and skin pigmentation. Pigment Cell Melanoma Res. 24, 401–410 (2011).
Ito, M., Kizawa, K., Hamada, K. & Cotsarelis, G. Hair follicle stem cells in the lower bulge form the secondary germ, a biochemically distinct but functionally equivalent progenitor cell population, at the termination of catagen. Differentiation 72, 548–557 (2004).
Paus, R., Sevilla, A. & Grichnik, J. M. Human hair graying revisited: principles, misconceptions, and key research frontiers. J. Investig. Dermatol. https://doi.org/10.1016/j.jid.2023.09.276 (2023).
Rompolas, P., Mesa, K. R. & Greco, V. Spatial organization within a niche as a determinant of stem-cell fate. Nature 502, 513–518 (2013).
Sun, Q. et al. Dedifferentiation maintains melanocyte stem cells in a dynamic niche. Nature 616, 774–782 (2023).
Clevers, H. The intestinal Crypt, a prototype stem cell compartment. Cell 154, 274–284 (2013).
Galvan, C. & Lowry, W. E. Yo-yoing stem cells defy dogma to maintain hair colour. Nature 616, 666–667 (2023).
Infarinato, N. R. et al. BMP signaling: at the gate between activated melanocyte stem cells and differentiation. Genes Dev. 34, 1713–1734 (2020).
Kubic, J. D., Young, K. P., Plummer, R. S., Ludvik, A. E. & Lang, D. Pigmentation PAX-ways: the role of Pax3 in melanogenesis, melanocyte stem cell maintenance, and disease. Pigment Cell Melanoma Res. 21, 627–645 (2008).
Lang, D. et al. Pax3 functions at a nodal point in melanocyte stem cell differentiation. Nature 433, 884–887 (2005).
Kawakami, A. & Fisher, D. E. The master role of microphthalmia-associated transcription factor in melanocyte and melanoma biology. Lab. Investig. 97, 649–656 (2017).
Palmer, J. W. et al. Molecular heterogeneity of quiescent melanocyte stem cells revealed by single-cell RNA-sequencing. Pigment Cell Melanoma Res. 37, 480–495 (2024).
Li, K. N. & Tumbar, T. Hair follicle stem cells as a skin-organizing signaling center during adult homeostasis. EMBO J. 40, e107135 (2021).
Massagué, J. TGFβ signalling in context. Nat. Rev. Mol. Cell Biol. 13, 616–630 (2012).
Nishimura, E. K. et al. Key roles for transforming growth factor beta in melanocyte stem cell maintenance. Cell Stem Cell 6, 130–140 (2010).
Matsumura, H. et al. Hair follicle aging is driven by transepidermal elimination of stem cells via COL17A1 proteolysis. Science 351, aad4395 (2016).
Tanimura, S. et al. Hair follicle stem cells provide a functional niche for melanocyte stem cells. Cell Stem Cell 8, 177–187 (2011).
Liu, N. et al. Stem cell competition orchestrates skin homeostasis and ageing. Nature 568, 344–350 (2019).
Yang, G. et al. Inhibition of PAX3 by TGF-beta modulates melanocyte viability. Mol. Cell 32, 554–563 (2008).
Czabotar, P. E., Lessene, G., Strasser, A. & Adams, J. M. Control of apoptosis by the BCL-2 protein family: implications for physiology and therapy. Nat. Rev. Mol. Cell Biol. 15, 49–63 (2014).
McGill, G. G. et al. Bcl2 regulation by the melanocyte master regulator Mitf modulates lineage survival and melanoma cell viability. Cell 109, 707–718 (2002).
Veis, D. J., Sorenson, C. M., Shutter, J. R. & Korsmeyer, S. J. Bcl-2-deficient mice demonstrate fulminant lymphoid apoptosis, polycystic kidneys, and hypopigmented hair. Cell 75, 229–240 (1993).
Nishimura, E. K., Granter, S. R. & Fisher, D. E. Mechanisms of hair graying: incomplete melanocyte stem cell maintenance in the niche. Science 307, 720–724 (2005).
Bray, S. J. Notch signalling in context. Nat. Rev. Mol. Cell Biol. 17, 722–735 (2016).
Borggrefe, T. & Oswald, F. The Notch signaling pathway: transcriptional regulation at Notch target genes. Cell Mol. Life Sci. 66, 1631–1646 (2009).
Lu, Z. et al. Hair follicle stem cells regulate retinoid metabolism to maintain the self-renewal niche for melanocyte stem cells. Elife 9, https://doi.org/10.7554/eLife.52712 (2020).
Niederreither, K. & Dollé, P. Retinoic acid in development: towards an integrated view. Nat. Rev. Genet. 9, 541–553 (2008).
Kumano, K. et al. Both Notch1 and Notch2 contribute to the regulation of melanocyte homeostasis. Pigment Cell Melanoma Res. 21, 70–78 (2008).
Moriyama, M. et al. Notch signaling via Hes1 transcription factor maintains survival of melanoblasts and melanocyte stem cells. J. Cell Biol. 173, 333–339 (2006).
Schouwey, K. et al. Notch1 and Notch2 receptors influence progressive hair graying in a dose-dependent manner. Dev. Dyn. 236, 282–289 (2007).
Aubin-Houzelstein, G. et al. Melanoblasts’ proper location and timed differentiation depend on Notch/RBP-J signaling in postnatal hair follicles. J. Investig. Dermatol. 128, 2686–2695 (2008).
Estrach, S., Ambler, C. A., Lo Celso, C. L., Hozumi, K. & Watt, F. M. Jagged 1 is a β-catenin target gene required for ectopic hair follicle formation in adult epidermis. Development 133, 4427–4438 (2006).
Nasrin, M. et al. Generation of Pmel-dependent conditional and inducible Cre-driver mouse line for melanocytic-targeted gene manipulation. Pigment Cell Melanoma Res. 36, 53–70 (2023).
Goding, C. R. & Arnheiter, H. MITF-the first 25 years. Genes Dev. 33, 983–1007 (2019).
Harris, M. L. et al. A dual role for SOX10 in the maintenance of the postnatal melanocyte lineage and the differentiation of melanocyte stem cell progenitors. PLoS Genet 9, e1003644 (2013).
Liu, J. et al. Wnt/β-catenin signalling: function, biological mechanisms, and therapeutic opportunities. Signal Transduct. Target Ther. 7, 3 (2022).
Ikeya, M., Lee, S. M. K., Johnson, J. E., McMahon, A. P. & Takada, S. Wnt signalling required for expansion of neural crest and CNS progenitors. Nature 389, 966–970 (1997).
Rabbani, P. et al. Coordinated activation of Wnt in epithelial and melanocyte stem cells initiates pigmented hair regeneration. Cell 145, 941–955 (2011).
Guo, H. et al. Wnt/β-catenin signaling pathway activates melanocyte stem cells in vitro and in vivo. J. Dermatol Sci. 83, 45–51 (2016).
Le Coz, M. et al. Targeted knockout of β-Catenin in adult melanocyte stem cells using a mouse line, Dct::CreERT2, results in disrupted stem cell renewal and pigmentation defects. J. Investig. Dermatol. 141, 1363–1366.e1369 (2021).
Choi, Y. eonS. et al. Distinct functions for Wnt/β-Catenin in hair follicle stem cell proliferation and survival and interfollicular epidermal homeostasis. Cell Stem Cell 13, 720–733 (2013).
Morris, R. J. et al. Capturing and profiling adult hair follicle stem cells. Nat. Biotechnol. 22, 411–417 (2004).
Tumbar, T. et al. Defining the epithelial stem cell niche in skin. Science 303, 359–363 (2004).
Guo, H. et al. Paracrine secreted frizzled-related protein 4 inhibits melanocytes differentiation in hair follicle. Stem Cells Int. 2017, 2857478 (2017).
Imokawa, G., Yada, Y. & Miyagishi, M. Endothelins secreted from human keratinocytes are intrinsic mitogens for human melanocytes. J. Biol. Chem. 267, 24675–24680 (1992).
Saldana-Caboverde, A. & Kos, L. Roles of endothelin signaling in melanocyte development and melanoma. Pigment Cell Melanoma Res. 23, 160–170 (2010).
Kim, T. H., Xiong, H., Zhang, Z. & Ren, B. beta-Catenin activates the growth factor endothelin-1 in colon cancer cells. Oncogene 24, 597–604 (2005).
Takeo, M. et al. EdnrB governs regenerative response of melanocyte stem cells by crosstalk with Wnt signaling. Cell Rep. 15, 1291–1302 (2016).
Becker-Santos, D. D., Lonergan, K. M., Gronostajski, R. M. & Lam, W. L. Nuclear factor I/B: A master regulator of cell differentiation with paradoxical roles in cancer. EBioMedicine 22, 2–9 (2017).
Chang, C.-Y. et al. NFIB is a governor of epithelial–melanocyte stem cell behaviour in a shared niche. Nature 495, 98–102 (2013).
Kato, M. et al. RET tyrosine kinase enhances hair growth in association with promotion of melanogenesis. Oncogene 20, 7536–7541 (2001).
Iida, M. et al. Hair graying with aging in mice carrying oncogenic RET. Aging Cell 19, e13273 (2020).
Lennartsson, J. & Rönnstrand, L. Stem cell factor receptor/c-Kit: from basic science to clinical implications. Physiol. Rev. 92, 1619–1649 (2012).
Botchkareva, N. V., Khlgatian, M., Longley, B. J., Botchkarev, V. A. & Gilchrest, B. A. SCF/c-kit signaling is required for cyclic regeneration of the hair pigmentation unit. Faseb J. 15, 645–658 (2001).
Liao, C. P., Booker, R. C., Morrison, S. J. & Le, L. Q. Identification of hair shaft progenitors that create a niche for hair pigmentation. Genes Dev. 31, 744–756 (2017).
Tandukar, B., Kalapurakal, E. & Hornyak, T. J. B6-Dct-H2BGFP bitransgenic mice: a standardized mouse model for in vivo characterization of melanocyte development and stem cell differentiation. Pigment Cell Melanoma Res. 34, 905–917 (2021).
Aoki, H., Tomita, H., Hara, A. & Kunisada, T. Conditional deletion of kit in melanocytes: white spotting phenotype is cell autonomous. J. Investig. Dermatol. 135, 1829–1838 (2015).
Kunisada, T. et al. Transgene expression of steel factor in the basal layer of epidermis promotes survival, proliferation, differentiation and migration of melanocyte precursors. Development 125, 2915–2923 (1998).
Nishimura, E. K. et al. Dominant role of the niche in melanocyte stem-cell fate determination. Nature 416, 854–860 (2002).
Cui, Y.-Z. et al. SPRY1 deficiency in keratinocytes induces follicular melanocyte stem cell migration to the epidermis through p53/stem cell factor/C-KIT signaling. J. Investig. Dermatol. https://doi.org/10.1016/j.jid.2024.02.018 (2024).
Vajravelu, B. N. et al. C-Kit promotes growth and migration of human cardiac progenitor cells via the PI3K-AKT and MEK-ERK pathways. PLOS ONE 10, e0140798 (2015).
Valluet, A. et al. B-Raf and C-Raf are required for melanocyte stem cell self-maintenance. Cell Rep. 2, 774–780 (2012).
Wang, R. N. et al. Bone Morphogenetic Protein (BMP) signaling in development and human diseases. Genes Dis. 1, 87–105 (2014).
Hsu, Y. C., Pasolli, H. A. & Fuchs, E. Dynamics between stem cells, niche, and progeny in the hair follicle. Cell 144, 92–105 (2011).
Plikus, M. V. et al. Cyclic dermal BMP signalling regulates stem cell activation during hair regeneration. Nature 451, 340–344 (2008).
Chen, C. C., Plikus, M. V., Tang, P. C., Widelitz, R. B. & Chuong, C. M. The modulatable stem cell niche: tissue interactions during hair and feather follicle regeneration. J. Mol. Biol. 428, 1423–1440 (2016).
Yue, Z., Jiang, T. X., Widelitz, R. B. & Chuong, C. M. Mapping stem cell activities in the feather follicle. Nature 438, 1026–1029 (2005).
Yue, Z., Jiang, T. X., Widelitz, R. B. & Chuong, C. M. Wnt3a gradient converts radial to bilateral feather symmetry via topological arrangement of epithelia. Proc. Natl. Acad. Sci. USA 103, 951–955 (2006).
Lin, S. J. et al. Topology of feather melanocyte progenitor niche allows complex pigment patterns to emerge. Science 340, 1442–1445 (2013).
Inaba, M. & Chuong, C. M. Avian pigment pattern formation: developmental control of Macro- (Across the Body) and micro- (Within a Feather) level of pigment patterns. Front. Cell Dev. Biol. 8, 620 (2020).
Shi, X. et al. Increased melanin induces aberrant keratinocyte − melanocyte − basal − fibroblast cell communication and fibrogenesis by inducing iron overload and ferroptosis resistance in keloids. Cell Commun. Signal. 23, 141 (2025).
Van Neste, D. & Tobin, D. J. Hair cycle and hair pigmentation: dynamic interactions and changes associated with aging. Micron 35, 193–200 (2004).
Endou, M., Aoki, H., Kobayashi, T. & Kunisada, T. Prevention of hair graying by factors that promote the growth and differentiation of melanocytes. J. Dermatol 41, 716–723 (2014).
Inoue-Narita, T. et al. Pten deficiency in melanocytes results in resistance to hair graying and susceptibility to carcinogen-induced melanomagenesis. Cancer Res. 68, 5760–5768 (2008).
Cavinato, M. & Jansen-Dürr, P. Molecular mechanisms of UVB-induced senescence of dermal fibroblasts and its relevance for photoaging of the human skin. Exp. Gerontol. 94, 78–82 (2017).
Aoki, H., Hara, A., Motohashi, T. & Kunisada, T. Protective effect of Kit signaling for melanocyte stem cells against radiation-induced genotoxic stress. J. Investig. Dermatol 131, 1906–1915 (2011).
Chou, W. C. et al. Direct migration of follicular melanocyte stem cells to the epidermis after wounding or UVB irradiation is dependent on Mc1r signaling. Nat. Med. 19, 924–929 (2013).
Inomata, K. et al. Genotoxic stress abrogates renewal of melanocyte stem cells by triggering their differentiation. Cell 137, 1088–1099 (2009).
Wolf Horrell, E. M., Boulanger, M. C. & D’Orazio, J. A. Melanocortin 1 receptor: structure, function, and regulation. Front. Genet 7, 95 (2016).
Yamada, T. et al. Wnt/β-catenin and kit signaling sequentially regulate melanocyte stem cell differentiation in UVB-induced epidermal pigmentation. J. Investig. Dermatol. 133, 2753–2762 (2013).
Herraiz, C. et al. Signaling from the human melanocortin 1 receptor to ERK1 and ERK2 mitogen-activated protein kinases involves transactivation of cKIT. Mol. Endocrinol. 25, 138–156 (2011).
Gorbunova, V., Seluanov, A., Mao, Z. & Hine, C. Changes in DNA repair during aging. Nucleic Acids Res. 35, 7466–7474 (2007).
Yu, M. et al. Deficiency in nucleotide excision repair family gene activity, especially ERCC3, Is associated with non-pigmented hair fiber growth. PLOS ONE 7, e34185 (2012).
Palmer, J. W. et al. Quiescence and aging of melanocyte stem cells and a novel association with programmed death-ligand 1. iScience 27, https://doi.org/10.1016/j.isci.2024.110908 (2024).
Rezayi, M. & Hosseini, A. Structure of PD1 and its mechanism in the treatment of autoimmune diseases. Cell Biochem. Funct. 41, 726–737 (2023).
Magomedova, A. & Fatima, G. Mental health and well-being in the modern era: a comprehensive review of challenges and interventions. Cureus 17, e77683 (2025).
Fujiwara, H. et al. The basement membrane of hair follicle stem cells is a muscle cell niche. Cell 144, 577–589 (2011).
Shwartz, Y. et al. Cell types promoting goosebumps form a niche to regulate hair follicle stem cells. Cell 182, 578–593.e519 (2020).
Rachmin, I. et al. Stress-associated ectopic differentiation of melanocyte stem cells and ORS amelanotic melanocytes in an ex vivo human hair follicle model. Exp. Dermatol. 30, 578–587 (2021).
Zhang, B. et al. Hyperactivation of sympathetic nerves drives depletion of melanocyte stem cells. Nature 577, 676–681 (2020).
Rosenberg, A. M. et al. Quantitative mapping of human hair greying and reversal in relation to life stress. eLife 10, e67437 (2021).
Feng, Z., Qin, Y. & Jiang, G. Reversing gray hair: inspiring the development of new therapies through research on hair pigmentation and repigmentation progress. Int. J. Biol. Sci. 19, 4588–4607 (2023).
Suzuki, T. et al. mTORC1 activity negatively regulates human hair follicle growth and pigmentation. EMBO Rep. 24, e56574 (2023).
Wu, S. et al. Single-cell transcriptomics reveals lineage trajectory of human scalp hair follicle and informs mechanisms of hair graying. Cell Discov. 8, 49 (2022).
Weiner, L. et al. Dedicated epithelial recipient cells determine pigmentation patterns. Cell 130, 932–942 (2007).
Enshell-Seijffers, D., Lindon, C., Kashiwagi, M. & Morgan, B. A. beta-catenin activity in the dermal papilla regulates morphogenesis and regeneration of hair. Dev. Cell 18, 633–642 (2010).
Cichorek, M., Wachulska, M., Stasiewicz, A. & Tymińska, A. Skin melanocytes: biology and development. Postepy Dermatol. Alergol. 30, 30–41 (2013).
Alam, M. et al. An osteopontin-derived peptide inhibits human hair growth at least in part by decreasing fibroblast growth factor-7 production in outer root sheath keratinocytes. Br. J. Dermatol. 182, 1404–1414 (2020).
Choi, H. I. et al. Hair greying is associated with active hair growth. Br. J. Dermatol. 165, 1183–1189 (2011).
Oh, J. W. et al. A guide to studying human hair follicle cycling in vivo. J. Investig. Dermatol. 136, 34–44 (2016).
Müller-Röver, S. et al. A comprehensive guide for the accurate classification of murine hair follicles in distinct hair cycle stages. J. Investig. Dermatol. 117, 3–15 (2001).
Olsen, W. M., Huitfeldt, H. S. & Eggset, G. UVB-induced (6-4) photoproducts in hairless mouse epidermis studied by quantitative immunohistochemistry. Carcinogenesis 10, 1669–1673 (1989).
Author information
Authors and Affiliations
Contributions
Conceptualization: T.O., D.L.G. Writing: T.O., D.L.G., and M.I. Paper and Data analyses: T.O., M.C., S.H.L., and D.L.G. Review and editing: T.O., D.L.G., and M.I. Visualization: T.O.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Ogawa, T., Chen, M., Lee, SH. et al. Dissecting the boundary of quiescence and activation of murine melanocyte stem cells in the hair follicle niche. npj Regen Med (2026). https://doi.org/10.1038/s41536-026-00460-3
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41536-026-00460-3