Abstract
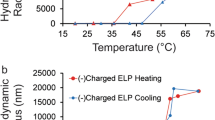
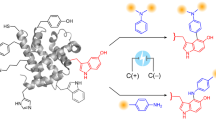
Elastin-like polypeptides (ELPs) are proteins naturally inspired by the hydrophobic domain of tropoelastin. ELPs are the repeating pentapeptide sequences of VPGXG (X can be any amino acid except proline) that undergo inverse transition behavior with temperature and salt stimuli. However, it remains challenging and unclear whether this transition, often accompanied by a conformational change, can be detected for short ELPs which are immobilized on surfaces. In this study, a sensing platform was developed based on the electrochemical activity of tyrosine to show conformational changes of newly designed, tethered, short ELPs with different hydrophobicity on gold working electrodes. Specifically, ELP sequences were modified with an N-terminal cysteine tag to attach to a solid gold surface via a thiol bond and modified with a C-terminal tyrosine tag, which can undergo electrochemical oxidation at the right potential. The ability of ELP conformational changes to modulate the oxidative current and indicate transition behavior was investigated via cyclic voltammetry. Additionally, the transition behavior of the short ELPs in solution was analyzed by UV-visible spectrometry for comparison. Overall, this study explores a distinct method for quantifying and studying the transition behavior of short, engineered peptides on a gold surface.
Similar content being viewed by others
Data availability
Data found in the main text of this manuscript can be found at: https://doi.org/10.7910/DVN/3D26NF.
References
Foster, J. A., Bruenger, E., Gray, W. R. & Sandberg, L. B. Isolation and amino acid sequences of tropoelastin peptides. J. Biol. Chem. 248, 2876–2879 (1973).
Urry, D. W. Entropic elastic processes in protein mechanisms. I. Elastic structure due to an inverse temperature transition and elasticity due to internal chain dynamics. J. protein Chem. 7, 1–34 (1988).
Gagner, J. E., Kim, W. & Chaikof, E. L. Designing protein-based biomaterials for medical applications. Acta Biomater. 10, 1542–1557 (2014).
Li, B., Alonso, D. O. & Daggett, V. The molecular basis for the inverse temperature transition of elastin. J. Mol. Biol. 305, 581–592 (2001).
Doberenz, F., Zeng, K., Willems, C., Zhang, K. & Groth, T. Thermoresponsive polymers and their biomedical application in tissue engineering–a review. J. Mater. Chem. B 8, 607–628 (2020).
Chilkoti, A., Christensen, T. & MacKay, J. A. Stimulus responsive elastin biopolymers: applications in medicine and biotechnology. Curr. Opin. Chem. Biol. 10, 652–657 (2006).
Nuhn, H. & Klok, H.-A. Secondary structure formation and LCST behavior of short elastin-like peptides. Biomacromolecules. 9, 2755–2763 (2008).
Urry, D. W. Molecular machines: how motion and other functions of living organisms can result from reversible chemical changes. Angew. Chem. Int. Ed. Engl. 32, 819–841 (1993).
Kowalczyk, T., Hnatuszko-Konka, K., Gerszberg, A. & Kononowicz, A. K. Elastin-like polypeptides as a promising family of genetically-engineered protein based polymers. World J. Microbiol. Biotechnol. 30, 2141–2152 (2014).
Wang, E. E. Development of Stimuli-Responsive Elastin-like Polypeptide-based Nanocomposite Biomaterials. (University of California, Berkeley and University of California, San Francisco, CA, 2012).
Despanie, J., Dhandhukia, J. P., Hamm-Alvarez, S. F. & MacKay, J. A. Elastin-like polypeptides: Therapeutic applications for an emerging class of nanomedicines. J. Controlled Rel. 240, 93–108 (2016).
Cook, W. J., Einspahr, H., Trapane, T. L., Urry, D. W. & Bugg, C. E. Crystal structure and conformation of the cyclic trimer of a repeat pentapeptide of elastin, cyclo-(L-valyl-L-prolylglycyl-L-valylglycyl) 3. J. Am. Chem. Soc. 102, 5502–5505 (1980).
Pechar, M. et al. Thermoresponsive Self-Assembly of Short Elastin-Like Polypentapeptides and Their Poly (ethylene glycol) Derivatives. Macromol. Biosci. 7, 56–69 (2007).
Flamia, R., Lanza, G., Salvi, A. M., Castle, J. E. & Tamburro, A. M. Conformational study and hydrogen bonds detection on elastin-related polypeptides using X-ray photoelectron spectroscopy. Biomacromolecules 6, 1299–1309 (2005).
Yao, X. & Hong, M. Structure distribution in an elastin-mimetic peptide (VPGVG) 3 investigated by solid-state NMR. J. Am. Chem. Soc. 126, 4199–4210 (2004).
Schreiner, E. et al. Folding and unfolding of an elastinlike oligopeptide:“inverse temperature transition,” reentrance, and hydrogen-bond dynamics. Phys. Rev. Lett. 92, 148101 (2004).
Reiersen, H., Clarke, A. R. & Rees, A. R. Short elastin-like peptides exhibit the same temperature-induced structural transitions as elastin polymers: implications for protein engineering. J. Mol. Biol. 283, 255–264 (1998).
Rousseau, R., Schreiner, E., Kohlmeyer, A. & Marx, D. Temperature-dependent conformational transitions and hydrogen-bond dynamics of the elastin-like octapeptide GVG (VPGVG): a molecular-dynamics study. Biophys. J. 86, 1393–1407 (2004).
Nicolini, C., Ravindra, R., Ludolph, B. & Winter, R. Characterization of the temperature-and pressure-induced inverse and reentrant transition of the minimum elastin-like polypeptide GVG (VPGVG) by DSC, PPC, CD, and FT-IR spectroscopy. Biophys. J. 86, 1385–1392 (2004).
Krukau, A., Brovchenko, I. & Geiger, A. Temperature-induced conformational transition of a model elastin-like peptide GVG (VPGVG) 3 in water. Biomacromolecules 8, 2196–2202 (2007).
Li, N. K., Quiroz, F. G., Hall, C. K., Chilkoti, A. & Yingling, Y. G. Molecular description of the LCST behavior of an elastin-like polypeptide. Biomacromolecules 15, 3522–3530 (2014).
Kurzbach, D. et al. Hydration layer coupling and cooperativity in phase behavior of stimulus responsive peptide polymers. J. Am. Chem. Soc. 135, 11299–11308 (2013).
MacEwan, S. R. & Chilkoti, A. Applications of elastin-like polypeptides in drug delivery. J. Controlled Rel. 190, 314–330 (2014).
Schweigerdt, A., Heinen, S., Stöbener, D. D. & Weinhart, M. Grafting density-dependent phase transition mechanism of thermoresponsive poly (glycidyl ether) brushes: a comprehensive QCM-D study. Langmuir 37, 7087–7096 (2021).
Su, Z., Kim, C. & Renner, J. N. Quantification of the effects of hydrophobicity and mass loading on the effective coverage of surface-immobilized elastin-like peptides. Biochem. Eng. J. 168, 107933 (2021).
Morales, M. A., Paiva, W. A., Marvin, L., Balog, E. R. M. & Halpern, J. M. Electrochemical characterization of the stimuli-response of surface-immobilized elastin-like polymers. Soft Matter 15, 9640–9646 (2019).
Dai, M. et al. Engineered protein polymer-gold nanoparticle hybrid materials for small molecule delivery. J. Nanomed. Nanotechnol. 7, 356 (2016).
Alvisi, N. et al. Self-assembly of elastin-like polypeptide brushes on silica surfaces and nanoparticles. Biomacromolecules 22, 1966–1979 (2021).
Ghoorchian, A., Cole, J. T. & Holland, N. B. Thermoreversible micelle formation using a three-armed star elastin-like polypeptide. Macromolecules 43, 4340–4345 (2010).
Christensen, T., Hassouneh, W., Trabbic-Carlson, K. & Chilkoti, A. Predicting transition temperatures of elastin-like polypeptide fusion proteins. Biomacromolecules 14, 1514–1519 (2013).
Pramounmat, N. Study of Elastin-Like Polypeptides Grafted on Electrode Surfaces (Case Western Reserve University, 2022).
Vanova, V. et al. Peptide-based electrochemical biosensors utilized for protein detection. Biosens. Bioelectron. 180, 113087 (2021).
Moulaee, K. & Neri, G. Electrochemical amino acid sensing: a review on challenges and achievements. Biosensors 11, 502 (2021).
Vestergaard, M. D. et al. A rapid label-free electrochemical detection and kinetic study of Alzheimer’s amyloid beta aggregation. J. Am. Chem. Soc. 127, 11892–11893 (2005).
Roeser, J., Permentier, H. P., Bruins, A. P. & Bischoff, R. Electrochemical oxidation and cleavage of tyrosine-and tryptophan-containing tripeptides. Anal. Chem. 82, 7556–7565 (2010).
Suprun, E. V. et al. Direct electrochemical oxidation of amyloid-β peptides via tyrosine, histidine, and methionine residues. Electrochem. Commun. 65, 53–56 (2016).
Suprun, E. V. et al. Tyrosine based electrochemical analysis of amyloid-β fragment (1-16) binding to metal (II) ions. Electrochim. Acta 179, 93–99 (2015).
Feeney, S. et al. Reproducibly modified elastin-like polymer gold electrode surfaces. ACS Meas. Sci. Au 5, 520–528 (2025).
Asaei, S., Verma, G., Sinclair, N. S. & Renner, J. N. Electrochemical biosensing of cerium with a tyrosine-functionalized EF-hand loop peptide. AIChE J. 70, e18620 (2024).
Hopp, T. P. & Woods, K. R. A computer program for predicting protein antigenic determinants. Mol. Immunol. 20, 483–489 (1983).
kyteKyte, J. & Doolittle, R. F. A simple method for displaying the hydropathic character of a protein. J. Mol. Biol. 157, 105–132 (1982).
Na, K. et al. Smart” biopolymer for a reversible stimuli-responsive platform in cell-based biochips. Langmuir 24, 4917–4923 (2008).
Garcia Quiroz, F. et al. Intrinsically disordered proteins access a range of hysteretic phase separation behaviors. Sci. Adv. 5, eaax5177 (2019).
Pramounmat, N. et al. Grafting of short elastin-like peptides using an electric field. Sci. Rep. 12, 1–13 (2022).
Marken, F., Neudeck, A. & Bond, A. M. Cyclic voltammetry. In Electroanalytical Methods: Guide to Experiments and Applications (ed. Scholz, F.), 57–106 (Springer Berlin Heidelberg, Berlin, Heidelberg, 2010).
Dinu, A. & Apetrei, C. Quantification of tyrosine in pharmaceuticals with the new biosensor based on laccase-modified polypyrrole polymeric thin film. Polymers 14, 441 (2022).
Harris, T. M., Devkota, J. P., Khanna, V., Eranki, P. L. & Landis, A. E. Logistic growth curve modeling of US energy production and consumption. Renew. Sustain. Energy Rev. 96, 46–57 (2018).
Kucharavy, D. & De Guio, R. Application of logistic growth curve. Proc. Eng. 131, 280–290 (2015).
Dreher, M. R. et al. Temperature triggered self-assembly of polypeptides into multivalent spherical micelles. J. Am. Chem. Soc. 130, 687–694 (2008).
de Haas, R. J., Ganar, K. A., Deshpande, S. & de Vries, R. pH-responsive elastin-like polypeptide designer condensates. ACS Appl. Mater. Interfaces 15, 45336–45344 (2023).
Ribeiro, A., Arias, F. J., Reguera, J., Alonso, M. & Rodríguez-Cabello, J. C. Influence of the amino-acid sequence on the inverse temperature transition of elastin-like polymers. Biophys. J. 97, 312–320 (2009).
Frey, W., Meyer, D. E. & Chilkoti, A. Thermodynamically reversible addressing of a stimuli responsive fusion protein onto a patterned surface template. Langmuir 19, 1641–1653 (2003).
Tang, J. D., Caliari, S. R. & Lampe, K. J. Temperature-dependent complex coacervation of engineered elastin-like polypeptide and hyaluronic acid polyelectrolytes. Biomacromolecules 19, 3925–3935 (2018).
Cho, Y. et al. Effects of Hofmeister anions on the phase transition temperature of elastin-like polypeptides. J. Phys. Chem. B 112, 13765–13771 (2008).
Reguera, J., Urry, D. W., Parker, T. M., McPherson, D. T. & Rodríguez-Cabello, J. C. Effect of NaCl on the exothermic and endothermic components of the inverse temperature transition of a model elastin-like polymer. Biomacromolecules. 8, 354–358 (2007).
Kunz, W., Nostro, P. L. & Ninham, B. W. The present state of affairs with Hofmeister effects. Curr. Opin. Colloid interface Sci. 9, 1–18 (2004).
Hostert, J. D. et al. Self-assembly and rearrangement of a polyproline II helix peptide on gold. Langmuir 37, 6115–6122 (2021).
Xia, C., Kang, W., Wang, J. & Wang, W. Temperature dependence of internal friction of peptides. J. Phys. Chem. B 125, 2821–2832 (2021).
Elgrishi, N. et al. A practical beginner’s guide to cyclic voltammetry. J. Chem. Educ. 95, 197–206 (2018).
Urry, D. W., Trapane, T. & Prasad, K. Phase-structure transitions of the elastin polypentapeptide–water system within the framework of composition–temperature studies. Biopolymers 24, 2345–2356 (1985).
Root, D. D., Yadavalli, V. K., Forbes, J. G. & Wang, K. Coiled-coil nanomechanics and uncoiling and unfolding of the superhelix and α-helices of myosin. Biophys. J. 90, 2852–2866 (2006).
Dixon, M. C. Quartz crystal microbalance with dissipation monitoring: enabling real-time characterization of biological materials and their interactions. J. Biomol. Tech. 19, 151 (2008).
Acknowledgements
This work was generously supported by the U.S. National Science Foundation (award number 2133549). We acknowledge Dr. Bridget Hegarty, assistant professor at the Department of Civil and Environmental Engineering of the Case School of Engineering, who let us use their MilliQ water system and Dr. Smarajit at Molecular Biotechnology Core housed in Lerner Research Building for letting us use the CD instrument.
Author information
Authors and Affiliations
Contributions
S.A. contributed to conceptualization, investigation, formal analysis, methodology, validation, visualization, project administration, writing - original draft, writing - review & editing; C.E.C. investigation, formal analysis, validation, visualization, writing - review & editing; E.I. formal analysis, validation, visualization, writing - review & editing; L.P. formal analysis, validation, visualization, writing - review & editing; N.S. conceptualization, investigation, formal analysis, validation, visualization, writing - review & editing; J.N.R. conceptualization, validation, visualization, formal analysis, funding acquisition, project administration, resources, supervision, writing - review & editing.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests
Peer review
Peer review information
Communications Chemistry thanks Diego Pallarola and the other, anonymous, reviewer(s) for their contribution to the peer review of this work.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Asaei, S., Couch, C.E., Ising, E. et al. Using cyclic voltammetry to probe the conformational transition of short elastin-like peptides. Commun Chem (2026). https://doi.org/10.1038/s42004-026-01987-8
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s42004-026-01987-8